Bioremediation is the use of biological agents, such as bacteria, fungi, or green plants, to remove or neutralize contaminants, as in polluted soil or water.

Bacteria and fungi generally work by breaking down contaminants such as petroleum into less harmful substances.
Toxicity
The degree to which a chemical substance or a particular mixture of substances can damage an organism.
Heavy metals
Heavy metals are naturally occurring elements that have a high atomic weight and a density at least 5 times greater than that of water.
Source of heavy metals
Heavy metals are naturally occurring elements that are found throughout the earth’s crust. Environmental contamination can also occur through metal corrosion, atmospheric deposition, soil erosion of metal ions and leaching of heavy metals, sediment re-suspension and metal evaporation from water resources to soil and ground water.
Natural phenomena such as weathering and volcanic eruptions have also been reported to significantly contribute to heavy metal pollution.
Industrial sources include metal processing in refineries, coal burning in power plants, petroleum combustion, nuclear power stations and high-tension lines, plastics, textiles, microelectronics, wood preservation and paper processing plant.
Metals such as cobalt (Co), copper (Cu), chromium (Cr), iron (Fe), magnesium (Mg), manganese (Mn), molybdenum (Mo), nickel (Ni), selenium (Se) and zinc (Zn) are essential nutrients that are required for various biochemical and physiological functions.
Heavy metals are also considered as trace elements because of their presence in trace concentrations (ppb range to less than 10ppm) in various environmental matrices.
Polluted Areas in Pakistan
Karachi is one of the most populated urban agglomerations in the world. The estimated baseline concentrations of Pb, Cr, Cu, Zn and Fe were 56.23, 12.9, 36.31, 123.03 and 11,776mg/kg, respectively.
In Islamabad total metal concentrations of Pb, Zn, Cu, Co, Ni, and Cr in soils varied between 2.0-29.0, 61.9-172.6, 8.9 to 357.4, 7.3-24.7, 41.4-59.3, and 40.2-927.2 mg/kg.
Lahore Is also affected by heavy metal contamination in soil and water. Concentration of Cd, Cu, Cr, Ni, Pb, Zn (environmental pollutants i.e. toxic elements), and Co, K, Mg, Mn, Na (common elements) in phosphate rocks used for production of fertilizer in Pakistan
Effects of heavy metals
In Plants
Toxic levels of heavy metal affect a variety of processes in plants (Maksymiec, 1997; Siedlecka et al., 2001). The most widely investigated metal is Cd.
Visible effects of exposure to high Cd2+ doses are growth inhibition and leaf chlorosis. The plant water balance is disturbed, stomatal opening is inhibited.
High Cd2+ doses also affect the photosynthetic apparatus. Several metabolic enzymes have been found to be sensitive to Cd. At the whole plant level Cu is an effective inhibitor of vegetative growth and induces general symptoms of senescence. Generation of ROS (reactive oxygen species) Substitution of essential metal ions by other incorrect ones.
In Humans
Some heavy metals (like Fe, Zn, Ca and Mg) have been reported to be of bio-importance to man and their daily medicinal and dietary allowances had been recommended.
However, some others (like As, Cd, Pb, and methylated forms of Hg) have been reported to have no known bio-importance in human biochemistry and physiology and consumption even at very low concentrations can be toxic.
The target organs for Cd toxicity have been identified as liver, placenta, kidneys, lungs, brain and bones. The consumption of heavy metal contaminated food can seriously deplete some essential nutrients in the body that are further responsible for decreasing immunological defenses, intrauterine growth retardation, disabilities associated with malnutrition and high prevalence of upper gastrointestinal.
Cancer rates Heavy metals uptake by plants and successive accumulation in human tissues and biomagnifications through the food chain causes both human health and environment concerns.
Heavy metals such as cadmium, lead and mercury are recognized reproductive toxicants, particularly for occupational workers with high levels of exposure (Järup, 2003; ATSDR, 2004).
Effects on soil
Soil contamination by heavy metals is of most important apprehension throughout the industrialized world. Heavy metal pollution also causes changes in the size, composition and activity of the microbial community.
The adverse effects of heavy metals on soil biological and biochemical properties are well documented. The soil properties i.e. organic matter, clay contents and pH have major influences on the extent of the effects of metals on biological and biochemical properties.
Heavy metals indirectly affect soil enzymatic activities by shifting the microbial community which synthesizes enzymes.
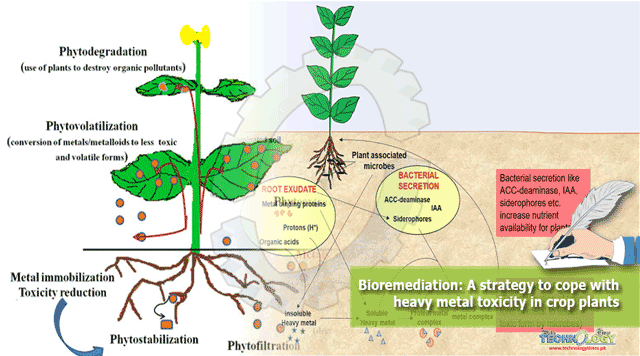
Phytoremediation
Phytoremediation is based on plant wich can extract and concentrate toxic element from contaminated soil (Raskin et al. 1997). It involves three mechanisms (Brooks et al. 1997, Baker and Brooks 1989)
Phytoextraction
Involving extraction of heavy metals from the soil; several wild plants concentrate high amounts of heavy metals in their
Phylofilteratio
Involving auatic and semiacuatic plants in various water purification system.
Phytovolatization
Involving volatization of some toxic metals like Se, As and Hg.
Species Hyperaccumulators of HMs
Some plant species have capacity to grow in the soil, contaminated with heavy metals and have capacity to accumulate elevated amount of heavy metals as an ecological adaptation in metalliferous soil (Shaw 1990, Maywald and weight 1997).
Brassica juncea was identified as valuable plant for removing heavy metal from soil. By Raskin et al. 1997. Paseolus vulgaris, poplar trees, Thlapsi goesingense, Armeria maritima, Arrhenatherum eletius, Arabidopsis hallerii, Festuca arunidinacea, Melilotus indica, and oat are the plant that considered as hyperaccumulated plant (R.K. Maiti et al. 2003)
Remarkably resistant plants (and organisms that constitute the plant’s rhizosphere) are involved in phytoremediation (consisting of phytoextraction, rhizofiltration and phytostabilization) of metal polluted sites.
Aerobic organisms have developed complex systems protecting them from ROS, consisting of several enzymes and antioxidants. Some external mechanisms that limit the uptake of metals by roots can help plants tolerate a certain amount of toxic metal in soil.
The ability of root cells to control the transport of heavy metals via membranes determines their tolerance by plants. In many cases plants resistant to heavy metal stress have lower nutritional requirements and specific mineral (cadmium, potassium and phosphorus) and water economies to cope with this stress.
Antioxidants
The most important antioxidant enzymes are: superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APO), monodehydroascorbate reductase (MDAR), and glutathione reductase (GTR).
Phenolic compounds
Phenolics have various functions in plants.
An enhancement of phenylopropanoid metabolism and the number of phenolic compounds can be observed under different environmental factors and stress conditions.
The synthesis of isoflavones and some other flavonoids is induced when plants are infected or injured. The induction of phenolic compound biosynthesis was observed in wheat in response to nickel toxicity and in maize in response to aluminum.
Phaseolus vulgaris exposed to Cd2+ accumulate soluble and insoluble phenolics and Phyllantus tenellus leaves contain more phenolics than control plants after being sprayed with copper sulphate. Increase in soluble phenolics in cell wall endurance and the creation of physical barriers preventing calls against harmful action of heavy metals.
Biochar
Several remediation techniques have been investigated to reduce the environmental impact of metal-contaminated soils, such as phytoremediation, soil washing, stabilisation, solidification, electroremediation and excavation (Mulligan et al., 2001; Kumpiene et al., 2008).
Agricultural, urban and industrial waste products can be used in soil remediation because these materials are typically inexpensive and available in large quantities (Lombi et al., 2002; Kumpiene et al., 2008).
In addition to diluting the concentration of the pollutant, these materials can reduce the mobility of heavy metals due to various mechanisms, such as adsorption, precipitation or complexation, decreasing the (bio)availability of pollutants in soils.
Biochars have also been studied as sorbents for inorganic and organic contaminants in water (Mohan et al., 2014) and some authors have been reported their use as amendments for soil remediation (Cao and Harris, 2010; Ahmad et al., 2014).
Organic materials can decrease the availability of heavy metals by increasing the soil pH and the specific interaction of metals because the reactive groups in the organic matter may complex and sorb the heavy metals present in the soil (Lee et al., 2004; Karlsson et al., 2007).
The use of compost originating from green, catering, paper and municipal solid waste significantly reduced the heavy metal concentration of Cu, Pb and Zn in a soil solution, but this effect was not observed for as (Farrell and Jones, 2010).
Different types of BC produced from herbaceous and woody biomass can be applied for HM immobilization and reduction of bioavailability in contaminated soils (Fan et al., 2012).
Yang X et al. (2016) applied BC derived from bamboo and rice straw (RS) to reduce the HM content with increasing application rates of BC and found that rice straw BC had greater potential, as compared to bamboo BC, and could significantly decrease the bioavailability of HMs, such as Cd, Cu, Pb, and Zn, in the soil. Puga et al. (2015) employed sugar cane straw-derived BC that decreased the availability of Cd, Pb, and Zn in mine polluted soils and the Cd, Pb, and Zn uptake by jack bean plants in an area with high levels of metals (especially Cd).
Biochar application to agricultural soils gradually promots microbial activity and plays vital roles in soil ecosystems, HM immobilization with dynamic key processes, such as organic matter breakdown, nutrient cycling, and crop production (Devare et al., 2007).
On the other hand, BC can change soil aeration and promote the arrangement of soil aggregates, as well as increase soil temperature (Grossman et al., 2010).
Microbes
Microbially catalyzed reactions, which occur in the natural sulfur cycle, have been integrated in a microbiological process to remove toxic metals from contaminated soils.
Bioleaching using sulfuric acid produced by sulfur-oxidizing bacteria was followed by precipitation of the leachate metals as insoluble sulfides by sulfate-reducing bacteria. By transfer of the yeast metallothionein gene into cauliflower increased Cd-tolerance and Cd accumulating ability can be conferred to the plant.
This article is written by Asad Shabbir department of Plant Breeding and Genetics, University of Agriculture Faisalabad, Pakistan.