Water pollution is a worldwide problem, affecting the health and life of humans, plants, and animals, along with the issue of freshwater scarcity.

Thus, considering the societal demands, environmental safety, and future sustainability, there is a dire need of tackling the challenges offered by water scarcity. It is estimated that 975,000 million gallons per day (MGD) wastewater produced including 674,000 MGD from municipal and 301,000 MGD from industrial uses. Owing to the scarcity and depletion of water, treatment, and reuse of wastewater have been become necessary for enhancing and conserving available freshwater resources. The treated water has several application areas including irrigation, aquaculture, industrial consumption, and groundwater recharge.
Depletion of Freshwater Resources
The world economy is based on just 0.5% of available freshwater resources which are depleting day by day. The rest of the water is confined to polar ice caps, glaciers, and oceans. About 70% of the available freshwater is under agriculture sector consumption for producing food and 30% is used by other consumers. Pakistan being an agricultural country needs to maintain its water resources which are decreasing with the increase in urbanization. Per capita, water availability has been dropped drastically to 935 m3 in 2020 from 5260 m3 in the last 70 years. So, there is a dire need to maintain the available water resources.
Instead of the fact that 70% part of the earth is inhabited by water, all of it is not available for maintaining life. A state’s economy is largely dependent on freshwater resources and it has always been the reason for conflicts among various nations. It is indispensable to agriculture and industry.
Nature of Pollutants in Wastewater:
The nature of water pollutants is dependent on the source of wastewater like household/domestic, or industrial. Domestic wastewater usually contains scum detergents, oil, vegetable residues, fuel, grease, and sediments. Industrial wastewater contains heavy metals, toxic chemicals, radioactive elements, organic wastes, acidic and basic wastes. All these pollutants have been classified into organic, inorganic, and biological ones on the basis of their nature. Among these pollutants, heavy metals are most perilous due to their persistent and bioaccumulation behavior. Bioaccumulation means the gathering of a substance in an organism in an amount greater than the concentration in the environment. Heavy metals tend to accumulate both in the environment and living organisms once they get in the system, instead of degradation. Organic matter is also found to be the most crucial water pollutant quantified by the total organic carbon. The source and nature of pollutants greatly influence the treatment processes involved in water decontamination. The figure below shows different stages of wastewater treatment.
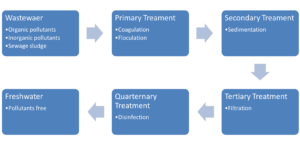
Techniques Available for Wastewater Treatment
Various processes present for the purification of wastewater are ion exchange, reverse osmosis, coagulation, chemical precipitation, ultrafiltration, electrolysis, and adsorption. The selection of the process depends upon the nature and concentration of the sorbent material used and the cost of treatment. Among these techniques, adsorption is the most useful and efficient one regarding supportive materials used in this technique, easy controlling, versatility, and cost-effectiveness. Adsorption is known to be a surface phenomenon and is widely useable for decontamination of all types of pollutants. It depends upon the interaction behavior of solid adsorbents having a porous structure and adsorbable solute.
The nature of adsorbents used in the treatment of wastewater is an important concern regarding its efficiency of pollutants removal. Traditionally used adsorbents include activated carbons, chelating materials, zeolites, and clay minerals. These adsorbents have the ability to remove toxic pollutants from wastewaters. But they have very low sorption capacity and application efficiency. In the last decade, a novel adsorbent has been introduced called “Nanoadsorbent” having a particle range of 1 to 100 nm to combat the deficiencies of conventional adsorbents.
Nano-adsorbents Properties
Nanoparticles are the most effective adsorbents owing to their very large surface areas, high surface binding energy, low-temperature adjustments, multiple sorption sites, large pore sizes, short diffusion range, and surface chemistry. The morphology of nanoparticles contributes a lot to their effectiveness and properties. It greatly affects the behavior of adsorption of toxic materials on the nano-adsorbent besides the composition and surface area. All these physical and chemical properties have made nano-adsorbents the most preferable materials for adsorption than the traditional sorbents. However, the distribution and nature of active sites, number, and types of functional groups help to show the adsorption behavior of nano-adsorbents.
Nano-adsorbent Types and Synthesis for Wastewater Treatment
Recent progress in the field of nanotechnology has open many ways to synthesize the nano-adsorbents like electrodeposition, laser ablation, solgel, catalytic growth, thermal spraying, ion sputtering, impregnation, spray pyrolysis, co-precipitation, sonochemical, arc discharge, thermal plasma synthesis, chemical vapor deposition, and mechanical milling from different sources including biomass residues and agricultural wastes.
Feasibility and Challenges Associated with Nano-adsorbents
The adoption feasibility and efficiency of a certain wastewater treatment technology depends upon two main factors including potential toxicity and cost-effectiveness. No doubt, nano-adsorbents offer very high pollutants removal efficiency but their cost and application are considered high. To overcome this issue low purity nano-adsorbents have been proposed for large-scale applications without reducing the adsorption rate. The separation (magnetic) and regeneration of nano-adsorbents make them more user-friendly and cost-effective. The application of nano-adsorbents for environmental remediation is the vastest use of nanoparticles. Although the economic benefits of nano-adsorbents are well known. But still, it requires safe and suitable strategies for the application of this technology to avoid the toxicities associated with it in wastewater treatment.
Conclusion
The use of nano-adsorbents for the treatment of wastewaters is among the most suitable techniques due to their effective surface binding properties. They can simultaneously remove the various types of pollutants which is the most challenging issue under the current scenario. So, it can be very helpful to treat the mixed waters sourced from different groups of the community.
This article is collectively authored by Muhammad Umair and Muhammad Saqib.