Parasites not only infect humans and animals but are a major burden on plants too. They are offering a deadly impact on livestock production, on the health of companion animals, on human health, and also on crop production.
By Dr. Maria Kausar
Introduction to Anthelmintic resistance
We are living in the 21st century, facing a major global issue, which is the development of resistance against parasitic drugs and the flourishing of the parasitic population on the whole globe. Anthelmintic resistance is the parasitic ability to survive against the recommended dose of any particular drug against that parasite which normally kills the parasites of the same species and same stage. It is a highly observed phenomenon these days. This is a very serious challenge for all of us to do something effective against this widespread parasitic population and day by day increasing resistance of these mini bodies. We have to make potentially fruitful approaches to combat this deadly scenario of resistance.
Ivermectin
Ivermectin is a well-known drug extensively used against parasites in humans as well as in animals. It is a semisynthetic analog of Avermectin (Abamectin) which is an insecticide developed for crop management. Notably, it is observed that ivermectin has been remained a novel drug both in human and veterinary medicine. This drug was recognized against animal parasites in 1981, and it became the highest-selling veterinary product in the world because it was equally effective against both ectoparasites and endoparasites. Now it is extensively used to control broad-spectrum infection which is caused by parasites and arthropods. Mass drug administration (MDA) strategy is used to control parasites in animals that are considered at risk. In humans, this strategy is used to control onchocerciasis and lymphatic filariasis. It reduces the intensity and prevalence of soil-borne helminths.

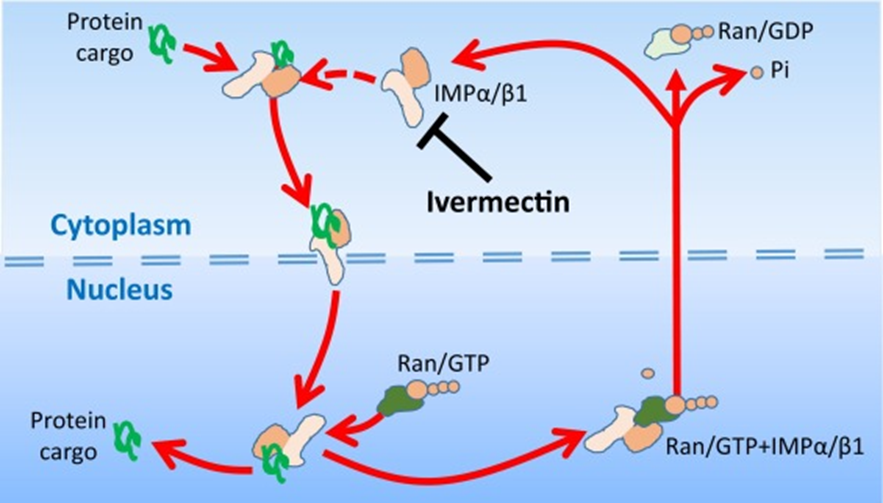
Ivermectin is reported to be effective against DNA as well as RNA viruses like Pseudorabies virus and West Nile virus, respectively. It inhibits virus replication and nowadays, it is being used massively against the causative agent of COVID-19, SARS CoV-2 that is a single standard positive-sense RNA virus. Resistance against parasites is developing due to over and irrational use of anthelmintics. There are many reported cases of ivermectin resistance. Ivermectin is extensively used in the feed formulation of salmon fish to control fish lice but resistance has also been developed in fish lice. It has been reported in GI parasites of camels and dogs. It has been reported into O. volvulus in response to ivermectin treatment in Sudan, Ghana, Cameroon, and other countries in humans.
Agro-environmental Impacts of Ivermectin
It is estimated that 49% of farmyard manure is used as organic fertilizer and is directly disposed of on lands to increase crop yield. Residues of veterinary parasiticides in the dung of treated livestock have a non-target effect on soil nematodes and dung breeding insects and indirectly affect dung degeneration. In this way, they are imposing environmental risks.
Mechanism of Resistance
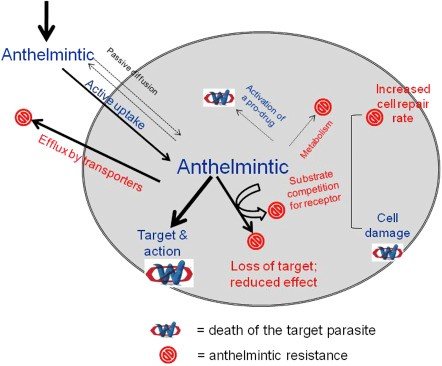
Drug metabolites mimic the parasites and pests to adapt, modify and transform them against this class of drug. Irrational use of drugs has led towards the lower efficacy of the drugs of the choice. If this resistance would go with the current ratio, then in near future there will be a shortage of meat production and indirectly affected the human population. The exact mechanism of resistance is unknown but it is elucidated that the parasites may develop certain membrane proteins or alteration in genes. Mutations in glutamate-gated chloride genes also induce resistance against ivermectin. There may be changes in glutamate-gated chloride channel receptor subunits. Such resistance puts great influences on public health as well as on livestock. In many farms, there is no proper management regarding deworming protocols, and a sublethal dose of ivermectin leads to rapid adaptation in parasites, and in this way, they are being immunized. There are different factors augmenting resistance in parasites include over-prescribing of ivermectin, farmers not giving doses as prescribed, unnecessary use in agriculture, lack of lab tests, and unavailability of the technical staff in the laboratory.
Control Strategy
Our livestock as well as the human population is in great threat, there is an urgent need to develop novel drugs or to ensure the rational use of available drugs like ivermectin. We have to make more targeted approaches to minimize this serious issue. Scientists are continuously introducing new techniques in drug development for the control of parasitic nematodes and other parasitic classes. Although resistance is an inevitable aspect of the use of anthelmintics. The general rule is that the more effective an anthelmintic is, the high chances of development of resistance, there will be. Parasites exhibit large genetic diversity of selection and adaptation to anthelmintics. Therefore, they are considered the most successful organisms on earth which is a serious threat to the other species. Judicious use of drugs is an integral part of clinical practices. It is an approach to maximize therapeutic efficacy and minimize the selection of resistant microorganisms. Chances of development of resistance will increase by increasing the frequency of treatment within the same environment. The use of lower doses of anthelmintics is another reason for this resistance. Farmers often underestimate the dose. A farmer must follow the drug dose rate and withdrawal period as recommended by the veterinarian. Another method to decrease the development of resistance is the rotation between the anthelmintics. They must be used from different classes rather than from a single class. Co-administration of anthelmintics also delays the chances of resistance in parasites as genes conferring resistance against both chemicals are rare in any individual nematode. There is a need to rapidly identify those genes that are responsible for creating resistance by using genetic markers. We will be able to identify the novel drug to counteract this issue not only in Pakistan but also in the world. There must be the discovery of a new molecular entity. Scientists in Pakistan must focus on this emerging issue for the promotion of human health as well as of animals.
References:
Hüter, O.F., 2011. Use of natural products in the crop protection industry. Phytochemistry reviews, 10(2), pp.185-194.
Amazigo U,Ann Trop Med Parasitol. 2008 Sep; 102 Suppl 1():19-22
Ottesen EA, Hooper PJ, Bradley M, Biswas G. The global programme to eliminate lymphatic filariasis: health impact after 8 years. PLoS Negl Trop Dis. 2008;2:e317.
Osei-Atweneboana MY, Eng JK, Boakye DA, Gyapong JO, Prichard RK (2007) Prevalence and intensity of Onchocerca volvulus infection and efficacy of ivermectin in endemic communities in Ghana: A two-phase epidemiological study. Lancet 369: 2021–2029.
Doyle, S.R., Bourguinat, C., Nana-Djeunga, H.C., Kengne-Ouafo, J.A., Pion, S.D., Bopda, J., Kamgno, J., Wanji, S., Che, H., Kuesel, A.C. and Walker, M., 2017. Genome-wide analysis of ivermectin response by Onchocerca volvulus reveals that genetic drift and soft selective sweeps contribute to loss of drug sensitivity. PLoS neglected tropical diseases, 11(7), p.e0005816.
Lv, C., Liu, W., Wang, B., Dang, R., Qiu, L., Ren, J., Yan, C., Yang, Z. and Wang, X., 2018. Ivermectin inhibits DNA polymerase UL42 of pseudorabies virus entrance into the nucleus and proliferation of the virus in vitro and vivo. Antiviral research, 159, pp.55-62.
Yang, S.N., Atkinson, S.C., Wang, C., Lee, A., Bogoyevitch, M.A., Borg, N.A. and Jans, D.A., 2020. The broad spectrum antiviral ivermectin targets the host nuclear transport importin α/β1 heterodimer. Antiviral research, 177, p.104760.
Makepeace, B.L., Babayan, S.A., Lustigman, S. and Taylor, D.W., 2015. The case for vaccine development in the strategy to eradicate river blindness (onchocerciasis) from Africa. Expert review of vaccines, 14(9), pp.1163-1165.
Hamed, M.I., 2018. Ivermectin resistance in intestinal parasites of camels in a private farm at Assiut, Egypt. Comparative Clinical Pathology, 27(5), pp.1221-1226.
Bamaiyi, P.H. and Kalu, A.U., 2011, January. Gastrointestinal parasites infection in one-humped camels (Camelus dromedarius) of Nigeria. VETERINARY RESEARCH FORUM.
Wolstenholme, A.J., Evans, C.C., D JIMENEZ, P.A.B.L.O. and Moorhead, A.R., 2015. The emergence of macrocyclic lactone resistance in the canine heartworm, Dirofilaria immitis. Parasitology, 142(10), p.1249.
E Horsberg, T., 2012. Avermectin use in aquaculture. Current pharmaceutical biotechnology, 13(6), pp.1095-1102.
www.biotechjournal.com/Journal/Mar2002/marArticle1text.htm
https://www.fda.gov/animal-veterinary/resources-you/ins-and-outs-extra-label-drug-use-animals-resource-veterinarians#animals
Dent, J.A., Smith, M.M., Vassilatis, D.K. and Avery, L., 2000. The genetics of ivermectin resistance in Caenorhabditis elegans. Proceedings of the National Academy of Sciences, 97(6), pp.2674-2679.
Prichard, R.K., 2007. Ivermectin resistance and overview of the Consortium for Anthelmintic Resistance SNPs. Expert Opinion on Drug Discovery, 2(sup1), pp.S41-S52.
Geary, T.G., Woo, K., McCarthy, J.S., Mackenzie, C.D., Horton, J., Prichard, R.K., de Silva, N.R., Olliaro, P.L., Lazdins-Helds, J.K., Engels, D.A. and Bundy, D.A., 2010. Unresolved issues in anthelmintic pharmacology for helminthiases of humans. International journal for parasitology, 40(1), pp.1-13.
Dash, K.M., Newman, R.L., Hall, E., 1985 Recommendations to minimise selection for anthelmintic resistance in nematode control programs. In (ed. N. Anderson and P.J. Waller) Resistance in nematodes to anthelmintic drugs. CSIRO, Glebe, NSW pp 161- 169
Prichard, R.K., Hall, C.A., Kelly, J.D., Martin, I.C.A., Donald, A.D., 1980. The problem of anthelmintic resistance in nematodes. Aust. Vet. J. 56, 239-250.
Barger, I.A., 1997. Models as a guide to sustainable worm control. In (ed G.K. Barrell) Sustainable control of internal parasites in ruminants. Animal Industries Workshop, Lincoln University, New Zealand, June 1997, pp 203-213
Kolar, L. and Eržen, N.K., 2006. Veterinary parasiticides–are they posing an environmental risk. Slov Vet Res, 43(2), pp.85-96.
Grønvold, J., Svendsen, T.S., Kraglund, H.O., Bresciani, J. and Monrad, J., 2004. Effect of the
antiparasitic drugs fenbendazole and ivermectin on the soil nematode Pristionchus
maupasi. Veterinary parasitology, 124(1-2), pp.91-99.
Authors:
- Dr. Maria Kausar, M.Phil. Parasitology, University of Agriculture, Faisalabad.
- Dr. Muhammad Sohail Sajid, Chairman Dept. of Parasitology at University of Agriculture Faisalabad.
- Rana M. Athar Ali, M.Phil. Clinical Medicine and Surgery, University of Agriculture Faisalabad.
- Arslan Nazim, DVM, University of Veterinary and Animal Sciences Lahore.